 In this review, authors focused on UC-MSC-based products based on their own experience. This required a rapid adaptation of stakeholders with the necessity to transfer this biotechnology from the “stem cell graft” status to the “drug” status. 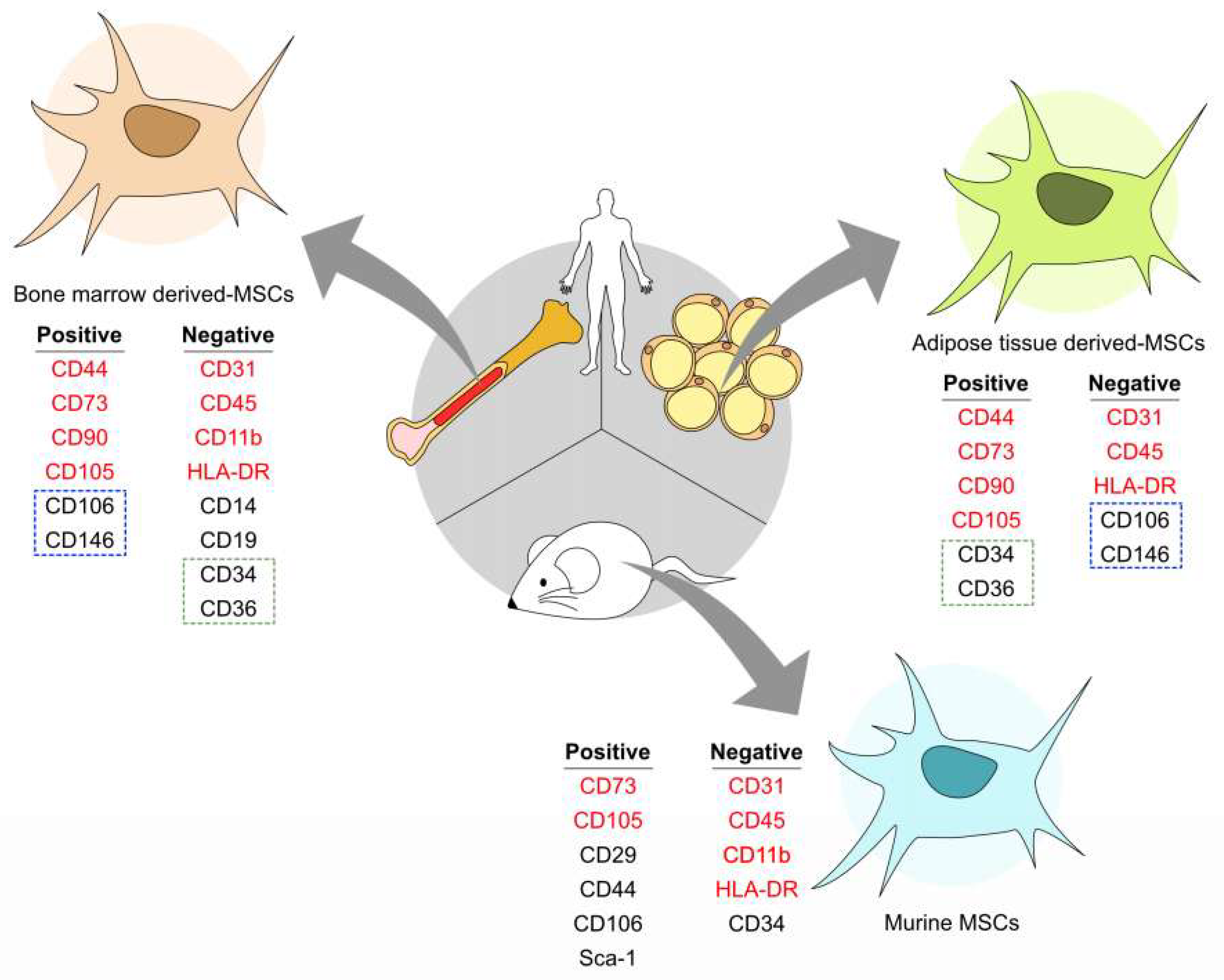
Indeed, since the publication of the European Regulation 1394/2007/EC introducing a new class of biological medicinal products, named advanced therapy medicinal products (ATMP), MSC-based products for therapeutic use must be developed according to this new status. It is important to notice that in this review authors considered only European framework, as they are cellular therapy experts regarding European regulation. The aim of our review is to point the perspective that offers UC-MSCs to develop innovative cellular medicines and then to discuss challenges of such products. However, no review has addressed the pharmaceutical development of UC-MSC-based medicines regarding regulatory aspects, manufacturing challenges and financial limits. These articles can be classified into five types: (i) describing UC-MSCs isolation, characterization, and biological properties, (ii) comparing UC-MSCs with MSCs from other tissues, (iii) describing UC-MSCs clinical potential in one specific disease or (iv) a systemic analysis of all clinical trials, and (v) describing exosomes derived from UC-MSCs. Only 90 reviews concerned UC-MSCs (reviews for: mesenchymal stem cell OR mesenchymal stromal cell AND umbilical cord OR Wharton jelly). Mesenchymal stem/stromal cells (MSCs) were widely described for several decades of years, with 3840 reviews referenced on the Medline/PubMed databases on November, 2020 (reviews for: mesenchymal stem cell OR mesenchymal stromal cell). Our aim was to point that despite MSCs use for several decades, the development of an UC-MSC-based ATMP remains at this day a real challenge for both academic institutions and pharmaceutical companies. Then, we discussed regulatory and manufacturing challenges for stakeholders, especially in terms of process harmonization and cells characterization. Despite the use of the same product, we noticed an important heterogeneity in terms of indication, posology and study design. After a brief description of UC-MSCs, we described in this review their recent use in a large panel of immune and inflammatory pathologies, including early and late phase clinical trials. Cell production in tissue and cell banks has been replaced by the manufacturing of a medicine, in authorized establishments, according to the good manufacturing practices (GMP) specific to ATMPs. 
This new regulatory status required a total adaptation of stakeholders wishing to develop UC-MSC-based ATMPs. Since 2007, UC-MSC-based products are classified as advanced therapy medicinal products (ATMP) according to the European Regulation 1394/2007/EC. Indeed, immunomodulatory and anti-inflammatory properties, associated to fewer ethical, availability, and safety issues, position UC-MSCs as a promising active substance to develop medicinal products. Umbilical cord-derived mesenchymal stem/stromal cells (UC-MSCs) emerge as a perspective for therapeutic use in immune and inflammatory diseases. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
